Pressrelease från CCHR International 2006-07-31
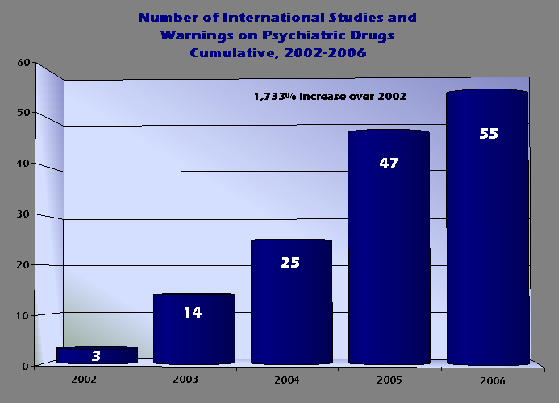
LATEST FDA WARNINGS ON ANTIDEPRESSANTS MARKS 1,700% INCREASE IN PSYCHIATRIC DRUG WARNINGS OVER LAST 4 YEARS
On July 21st, the U.S. Food and Drug Administration (FDA) asked antidepressant drug makers to add two more warnings to the drug labels. According to the FDA, antidepressants such as Paxil, Prozac or Zoloft taken during pregnancy can cause the newborn to develop a fatal lung disease. The FDA also warned that taking antidepressants with certain migraine drugs can lead to "serotonin syndrome," which can lead to hallucinations, vomiting, blood pressure changes, restlessness and death. These two most recent warnings about antidepressants may sound shocking, but they are consistent with the disturbing history of psychiatric drug warnings.
The following is a list of all Psychiatric Drug Warnings Issued Since 2004
 2004
February 2: FDA official Dr. Andrew D. Mosholder testified before the FDA s Psychopharmacological Advisory Committee on the Office of Drug Safety Data Resources for the Study of Suicidal Events, warning that children being prescribed the newer antidepressants were at risk of suicide.
March 22: The FDA warned that Prozac-like antidepressants (called Selective Serotonin Reuptake Inhibitors or SSRIs) could cause "anxiety, agitation, panic attacks, insomnia, irritability, hostility, impulsivity, akathisia [severe restlessness], hypomania [abnormal excitement] and mania [psychosis characterized by exalted feelings, delusions of grandeur]."
June: The Australian Therapeutic Goods Administration published an Adverse Drug Reactions Bulletin reporting that the latest antipsychotics could increase the risk of diabetes.
June: The FDA ordered that the packaging for the stimulant Adderall include a warning about sudden cardiovascular deaths, especially in children with underlying heart disease.
August 20: A Columbia University review of the pediatric (child) clinical trials of Zoloft, Celexa, Effexor, Wellbutrin, Paxil and Prozac, found that young people who took them could experience suicidal thoughts or actions.
September 21: Following a BBC news report on antidepressants causing aggression and homicidal behavior, the British Healthcare Products Regulatory Authority advised that it had issued guidelines that children should not be given most SSRIs because clinical trial data showed an increased rate of harmful outcomes, including hostility.
October 15: The FDA ordered a "black box" warning for antidepressants that they could cause suicidal thoughts and actions in under 18 year olds taking them.
October 21: The New Zealand Medicines Adverse Reactions Committee recommended that old and new antidepressants not be administered to patients less than 18 years of age because of the potential risk of suicide.
December: The Australian Therapeutic Goods Administration children and adolescents prescribed SSRI antidepressants should be carefully monitored for the emergence of suicidal ideation. In a recent study involving Prozac, it said, there was an increase in adverse psychiatric events (acts and ideation [thoughts] of suicide, self-harm, aggression, violence.)
December 9: The European Medicines Agency s Committee for Medicinal Products for Human Use confirmed that product information should be changed for antidepressants to warn of the risk of suicide-related behavior in children and adolescents and of withdrawal reactions on stopping treatment.
December 17: The FDA required that packaging for the "ADHD" drug Straterra carry a new warning advising, "Severe liver damage may progress to liver failure resulting in death or the need for a liver transplant in a small percentage of patients."
2005
February 9: Health Canada, the Canadian counterpart of the FDA, suspended marketing of Adderall XR (Extended Release, given once a day) due to reports of 20 sudden unexplained deaths (14 in children) and 12 strokes (2 in children) in patients taking Adderall or Adderall XR.
February 18: A study published in the British Medical Journal determined that adults taking SSRI antidepressants were more than twice as likely to attempt suicide as patients given placebo.
April: The British House of Commons (Parliament) Health Committee issued a damning report that SSRI antidepressants had been "indiscriminately prescribed on a grand scale" and that drug companies have marketed the drugs without punishment to treat "unhappiness [that] is part of the spectrum of human experience, not a medical condition."
April 11: The FDA warned that antipsychotic drugs in elderly patients could increase the risk of death.
April 21: A national non-government organization, Partnership for a Drug-Free America, released its findings of a study that determined that 10% of teens (2.3 million) had abused the stimulants Ritalin and Adderall.
April 25: The European Medicines Agency s Committee for Medicinal Products for Human Use reaffirmed that all the latest antidepressants could cause increased suicide-related behavior and hostility in young people.
June 28: The FDA announced its intention to make labeling changes to Concerta and other Ritalin products to include the side effects: "visual hallucinations, suicidal ideation [ideas], psychotic behavior, as well as aggression or violent behavior."
June 30: The FDA warned that the latest antidepressant Cymbalta could increase suicidal thinking or behavior in pediatric patients taking it.
June 30: The FDA also warned about a potential increased risk of suicidal behavior in adults taking antidepressants, broadening its earlier warning that related only to children and adolescents taking the drugs.
July 1: An FDA "Talk Paper" said that it had requested antidepressant manufacturers to provide all information from their clinical trials on possible increased suicidal behavior in adults taking the drugs.
July 7: The National Center on Addiction and Substance Abuse at Columbia University issued a report called "Under the Counter: The Diversion and Abuse of Controlled Prescription Drugs in the U.S." that determined 15 million Americans were getting high on painkillers and psychiatric drugs such as the tranquilizer Xanax and the stimulants Ritalin and Adderall. Between 1992 and 2003, the number of 12 to 17 year olds abusing these drugs had risen 212%. Teens who abused prescription drugs were 12 times likelier to use heroin, 15 times likelier to use Ecstasy and 21 times likelier to use cocaine, compared to teens that did not abuse such drugs.
July 16: The British Medical Journal published a study, "Efficacy of antidepressants in adults," by Joanna Moncrieff, senior lecturer in psychiatry at University College London who found that antidepressants were no more effective than a placebo (fake pill) and do not reduce depression. In a media interview on the study, Dr. Moncrieff stated, "The bottom line is that we really don t have any good evidence that these drugs work."
August: The Australian Therapeutic Goods Administration found a relationship between antidepressants and suicidality, akathisia (severe restlessness), agitation, nervousness and anxiety in adults. Similar symptoms could occur during withdrawal from the drugs, it determined.
August 19: The European Medicines Agency s Committee for Medicinal Products for Human Use issued its strongest warning against child antidepressant use, stating that the drugs caused suicide attempts and thoughts, aggression, hostility, aggression, oppositional behavior and anger.
August 22: Norwegian researchers found that patients taking antidepressants were seven times more likely to experience suicide than those taking placebo.
September 7: The Australian Therapeutic Goods Administration warned that antidepressant use during pregnancy could cause "withdrawal effects that can be severe or life-threatening."
September 13: The Oregon Health & Science University, Evidence-Based Practice Center published the findings of its review of 2,287 studies virtually every study ever conducted on "ADHD" drugs and found that there were no trials showing the effectiveness of these drugs and that there was a lack of evidence that they could affect "academic performance, risky behaviors, social achievements, etc." Further, "We found no evidence on long-term safety of drugs used to treat ADHD in young children" or "adolescents."
September 22: Dr. Jeffrey Lieberman of Columbia University and other researchers published a federally funded study in the New England Journal of Medicineabout the effectiveness of certain antipsychotic drugs, comparing an older generation of antipsychotics with several newer ones. Far from proving effectiveness, of the 1,493 patients who had participated, 74% discontinued their antipsychotic drugs before the end of their treatment due to inefficacy, intolerable side effects or other reasons. After 18 months of taking Zyprexa, 64% of the patients taking this stopped, most commonly because it caused sleepiness, weight gain or neurological symptoms like stiffness and tremors.
September 26: The Italian Gazette (official news agency of the Italian government) published a resolution of the Agenzia Italiana del Farmaco (Italian Drug Agency, equivalent to the FDA) ordering a warning label for older (tricyclic) antidepressants that the drugs should not be prescribed for under 18 year olds. They also determined that they were associated with heart attacks in people of any age.
September 27: The FDA warned that Paxil and other antidepressants taken during the first trimester of pregnancy could cause increased risk of major birth defects, including heart malformations in newborn infants.
September 28: The British National Health Service s Institute for Health and Clinical Excellence released a Clinical Guideline for treatment of "Depression in Children and Young People." It advised "all antidepressant drugs have significant risks when given to children and young people" and instead, they should be "offered advice on the benefits of regular exercise," "sleep hygiene," "nutrition and the benefits of a balanced diet."
September 29: The FDA directed Eli Lilly & Co. to revise Strattera labeling to include a boxed warning about the increased risk of suicidal thinking in children and adolescents taking it.
September 29: The UK Medicines and Healthcare Products Regulatory Agency issued a press release that it had begun a review of the risks of Strattera in light of the FDA s direction.
October: The sales and marketing of the stimulant Cylert were stopped in the U.S. because of the risk of liver damage that could lead to death.
October 17: The FDA ordered Eli Lilly & Co. to add a warning to the packaging of its antidepressant Cymbalta, that it could cause liver damage.
October 19: A study in the Journal of the American Medical Association concluded that atypical (newer) antipsychotic drugs could increase the risk of death in elderly people.
October 24: The FDA withdrew Cylert from the market because of its "overall risk of liver toxicity" and liver failure.
November: The FDA approved updated labeling for the antidepressant Effexor XR which noted that this antidepressant can cause homicidal ideation.
December 1: Researchers found that 18% of nearly 23,000 elderly patients taking the older antipsychotics died within the first six months of taking them.
December 8: The FDA warned that Paxil taken by pregnant women in their first trimester may cause birth defects, including heart malformations.
2006
January 5: The FDA said it had received reports of sudden deaths, strokes, heart attacks and hypertension (high blood pressure) in both children and adults taking "ADHD" drugs and asked its Drug Safety and Risk Management advisory committee to examine the potential of cardiovascular (heart) risks of the drugs.
February 4: A University of Texas study published in Pediatric Neurology reported cardiovascular problems in people taking stimulants.
February 5: An analysis of World Health Organization medical records found that infants whose mothers took antidepressants while pregnant could suffer withdrawal effects.
February 6: A study published in the Archives of Pediatrics and Adolescent Medicine determined that nearly one-third of newborn infants whose mothers took SSRI antidepressants during pregnancy experienced withdrawal symptoms that included high-pitched crying, tremors and disturbed sleep.
February 9: The FDA s Drug Safety and Risk Management Advisory Committee urged that the strongest "black box" warning be issued for stimulants, including Ritalin, Adderall and Concerta and that they may cause heart attacks, strokes and sudden death.
February 11: The Australian Therapeutic Goods Administration announced it would review the FDA advisory committee recommendation for stronger warnings against stimulants.
February 20: British authorities warned that the "ADHD" drug Strattera was associated with seizures and potentially lengthening period of the time between heartbeats.
February 25: A study in the journal, Drug and Alcohol Dependence, and reported in The Washington Post revealed that seven million Americans were estimated to have abused stimulant drugs and a substantial amount of teenagers and young adults now appeared to show signs of addiction.
March 10: Health Canada issued a warning that pregnant women taking SSRIs and other newer antidepressants placed newborns at risk of developing a rare lung and heart condition.
March 22-23: Two FDA advisory panels held hearings into the risk of stimulants and another new "ADHD" drug called Sparlon. Between January 2000 and June 30, 2005, the FDA had received almost 1,000 reports of kids experiencing psychosis or mania while taking the drugs. The first panel recommended stronger warnings against stimulants, emphasizing these on special handouts called "Med Guides" that doctors must give to patients with each prescription. The second committee recommended not to approve Sparlon, which the manufacturer, Cephalon, estimated would lose them $100 million in drug sales.
March 28: The Australian Therapeutic Goods Administration announced its review of reports of 400 adverse reactions to stimulants in children taking them. CCHR had filed a Freedom of Information Act request with the TGA to obtain the reports and released this to the media that ran the story internationally.
May 1: An American Journal of Psychiatry study revealed that elderly people prescribed antidepressants such as Prozac, Paxil, and Zoloft are almost five times more likely to commit suicide during the first month on the drugs than those given other classes of antidepressants.
May 3: FDA adverse drug reaction reports linked 45 child deaths to new antipsychotic drugs. There were also more than 1,300 reports of other potentially life-threatening adverse reactions such as convulsions and low white blood cell count.
May 12: GlaxoSmithKline, the manufacturer of Paxil, sent a letter to doctors warning that its antidepressant increases the risk of suicide in adults. It was the first warning of its kind by a manufacturer.
July 19: The FDA said antidepressant packaging should carry warnings that they may cause a fatal lung condition in newborns whose mothers took SSRI antidepressants during pregnancy. Migraine sufferers also need to be warned that combining migraine drugs with SSRIs could result in a life-threatening condition called serotonin syndrome. |